Increasingly in recent years, the NHS has struggled to buy drugs people need to manage mental ill health and other common conditions like heart disease at normal prices. As well as costing the health service hundreds of millions in unexpected costs, where this reflects underlying shortages patients risk finding the medicine they need is out of stock in their local pharmacies.
More than 80% of the medicines purchased for prescribing by general practices and dispensing by pharmacies are generic – off-patent drugs that any pharmaceutical company can manufacture and market. Where these drugs are not easily available, perhaps as a result of manufacturing issues or a steep change in demand, the NHS may decide to secure supplies by adding them to a ‘concessionary’ list for a time and paying a higher price.
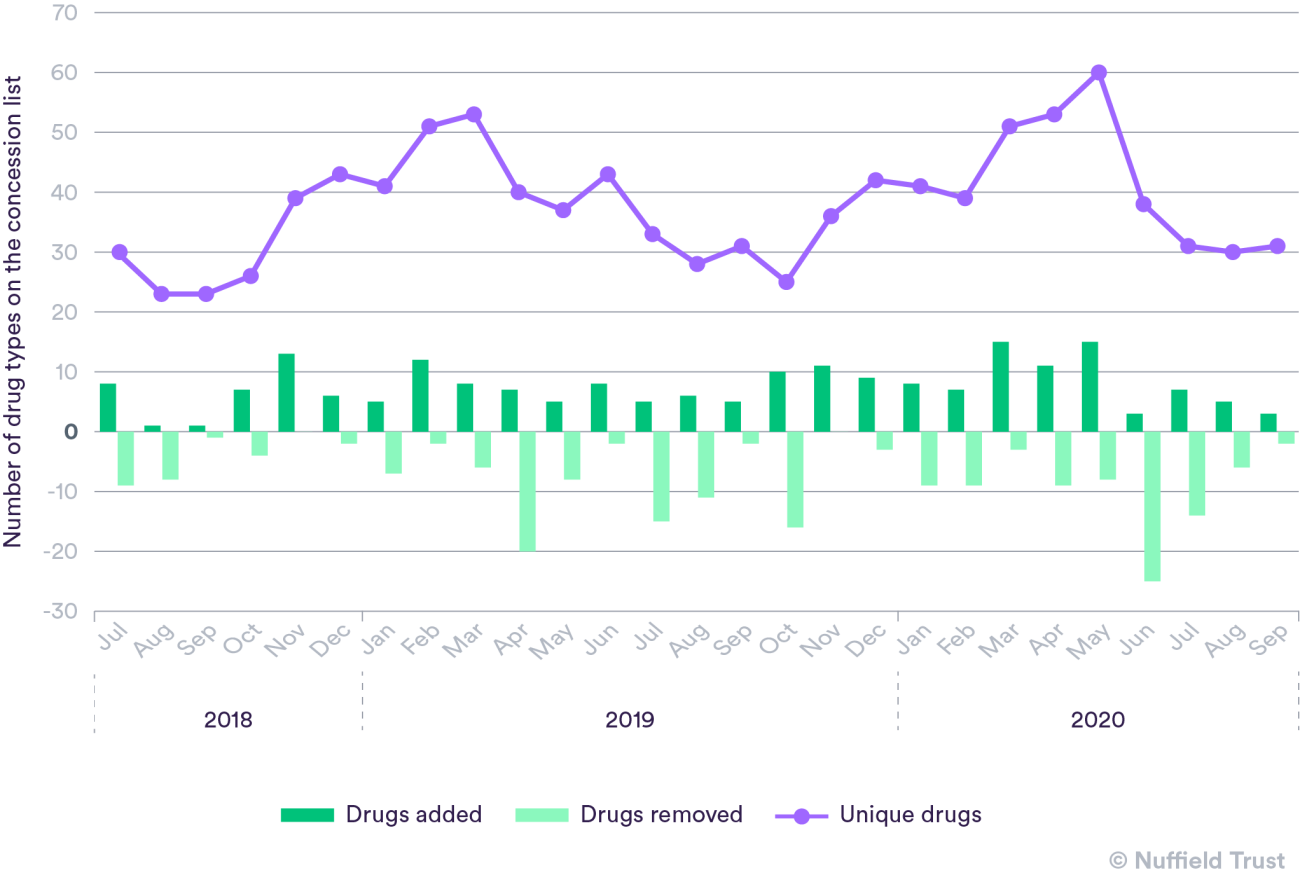
Around the start of the Covid-19 pandemic there was a rise in the number of drugs added to the price concession list. As well as items such as paracetamol and ibuprofen, which many people struggled to purchase themselves in the first three months of the pandemic, drugs used by health care professionals to treat a variety of conditions became hard to access at the standard price. These included drugs used to manage anxiety and depression, treatment for epilepsy, drugs used to lower cholesterol, and pain relief medication. The number of drugs on the price concession list peaked in May 2020, with 60 unique drug types. This was the highest number of unique drugs on this list in more than two-and-a-half years.
In the face of increasing cases of Covid-19 we may expect to see a rise in the number of drugs necessitating a higher cost price over the coming months. As well as being affected by the pandemic, drug access challenges may be further compounded as we move out of the Brexit transition period at the end of December.
Data notes
- The data source is the Pharmaceutical Services Negotiating Committee price concession and NCSO archive list.
- We looked at unique drug types on each monthly list, meaning that where a drug/medicine was on the list with multiple package sizes or formulations it was only counted once.
- Data covers the time period from July 2018 to September 2020. July 2018 was chosen as the starting point as this is when new rules came in to help regulate generic drug pricing.
- The graph shows the total number of unique drugs on the list by month as well as how many new drugs were added each month, and how many drugs had been removed from the list since the previous month.